Rucete ✏ Chemistry In a Nutshell
Alkane
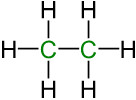
The alkanes are components of a homologous series of saturated hydrocarbons with the general formula Cn H2n+2. Also, cyclic alkanes with the general formula Cn H2n.
How to Name?
The alkanes are identified by the correct prefix for the amount of carbons followed by the suffix '-ane.'
Eth /ane (two carbons / alkane) = C2H6
Physical characteristics
- They do not combine with water because they are non-polar molecules; consequently, two layers form, with the alkane float on top because it is less dense. Alkanes can be dissolved by non - polar solvents.
- Smaller alkanes (1–4 carbons) are gases, medium-sized alkanes (5–16 carbons) are liquids, and larger alkanes are waxy solids.
- Alkanes just aren't electrically conductive.
- Branched chain alkanes have lower boiling and melting points than unbranched compounds. Because branches take up more space, branched molecules have greater space between them, which reduces intermolecular forces.
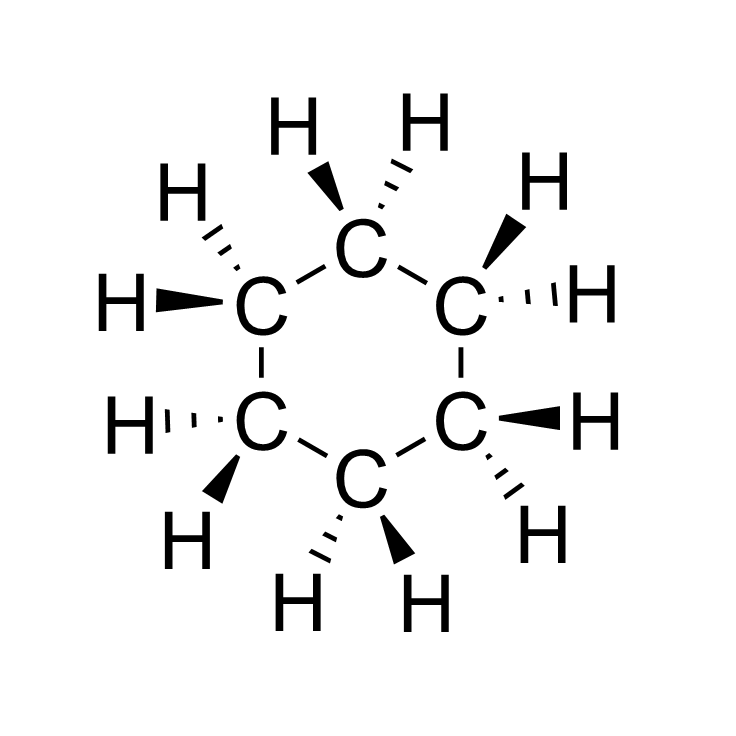
- Cyclic alkane
Cyclic alkanes are saturated hydrocarbons with ring-structured carbons. Cyclic alkanes are significantly lighter than straight chain alkanes as well as boil at 10-20°C higher temperatures.