Rucete ✏ Chemistry In a Nutshell
Alkenes
The alkenes are part of a homologous series of unsaturated hydrocarbons with the general formula CnH2n.
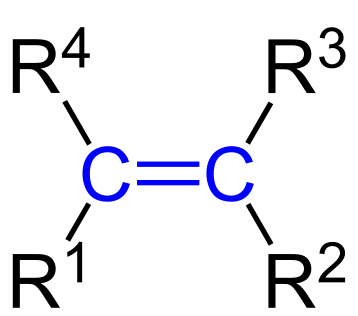
Alkenes have a double bond of the type C=C.
How to name?
An alkene is named by replacing the ‘-ane’ of the corresponding alkane with ‘-ene’. When required the position of the double bond must be specified.
Also, the position of the double bond must be indicated when necessary.
Physical characteristics
- As with alkanes, the melting and boiling temperatures increase as the number of carbon atoms or molar mass increases due to an increase in transient dipole-dipole attraction.
- Since alkenes are non-polar, they are insoluble in polar solvents such as water. Conversely, they are soluble in non-polar solvents.