Rucete ✏ Chemistry In a Nutshell
Chemical properties of Alkanes
The alkanes are homologous to a series of saturated hydrocarbons with the general formula CnH2n+2. Cyclic alkanes with the general formula CnH2n are also present.
In comparison to other functional groups, alkanes are relatively inactive. They are insoluble in acids and oxidizing agents. Their reactions require ultraviolet light or incredibly high temperatures to initiate. Cyclic alkanes have a higher reactivity than straight chain of these.
Substitution
Alkanes undergo a substitution reaction with halogens. This requires ultraviolet light or extremely high temperatures.
Alkane + Halogen --(UV)--> Haloalkane + Hydrogen halide
Example: CH4 + Cl2 --(UV)--> CH3Cl + HCl
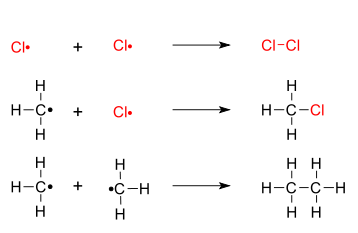
(Mechanism)
Cl -> 2Cl
Cl + CH4 -> HCl+ CH3
CH3 + Cl2 -> CH3Cl + Cl
 |
| Example of cyclic alkanes (cyclohexane) |
Combustion
Burning alkanes produces carbon dioxide and water vapor in abundance of oxygen.
Additionally, when oxygen is limited, carbon and carbon monoxide are produced.
Alkane + O2 (g) --(burning)--> CO2(g) + H2O(g)
E.g. Combustion of butane
2C4H10 + 13O2(g)--> 8CO2(g) + 10H2O(g)